TL;DR:
- X-ray absorption spectroscopy provides element-specific local atomic and electronic environment details unmatched by XRD or XRF.
- Proper experimental design, calibration, and complementary techniques are essential to avoid misinterpretation of XAS data.
- Combining operando XAS with AI and other analytical methods enables detailed insights into dynamic structural changes in complex systems.
X-ray techniques are often grouped together as if interchangeable, but that assumption leads research teams to choose the wrong tool and draw incomplete conclusions. X-ray absorption spectroscopy measures the absorption coefficient as a function of photon energy near an element’s absorption edge, probing local atomic structure and electronic environment with a specificity no other routine X-ray method matches. Whether your work involves tracking oxidation states in battery cathodes, characterizing protein-metal coordination in drug candidates, or resolving phase transformations in engineering alloys, XAS delivers information that XRD and XRF simply cannot. This article covers the foundational principles, detection modes, practical case studies, and critical pitfalls your team needs to know.
Table of Contents
- What is x-ray absorption spectroscopy (XAS)?
- How XAS works: Modes, energies, and detection
- Interpreting XAS data: Case studies and benchmarks
- Real-world applications: Pharmaceutical and materials breakthroughs
- Nuances and pitfalls: Advanced considerations in XAS
- Why most XAS interpretations fall short—and how to get it right
- Unlock the full power of XAS in your research
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Local structure insight | XAS uniquely measures an element’s atomic environment, revealing details missed by XRD or XRF. |
| Versatile sample analysis | You can obtain information from solid, liquid, or even in-operando cells using various detection modes. |
| Beware interpretation pitfalls | Account for beam damage, self-absorption, and ensure use of standards to avoid artifacts in your results. |
| Critical for innovation | XAS accelerates breakthroughs in pharma, biomedicine, and advanced materials research. |
| Integrate with computation | Combining XAS data with theory or machine learning maximizes reliability and discovery. |
What is x-ray absorption spectroscopy (XAS)?
XAS measures how strongly a material absorbs X-rays as photon energy is scanned across the absorption edge of a target element. When photon energy matches the binding energy of a core electron, absorption rises sharply, creating the characteristic “edge.” That edge and the oscillations surrounding it carry detailed information about the local chemical environment of the absorbing atom.
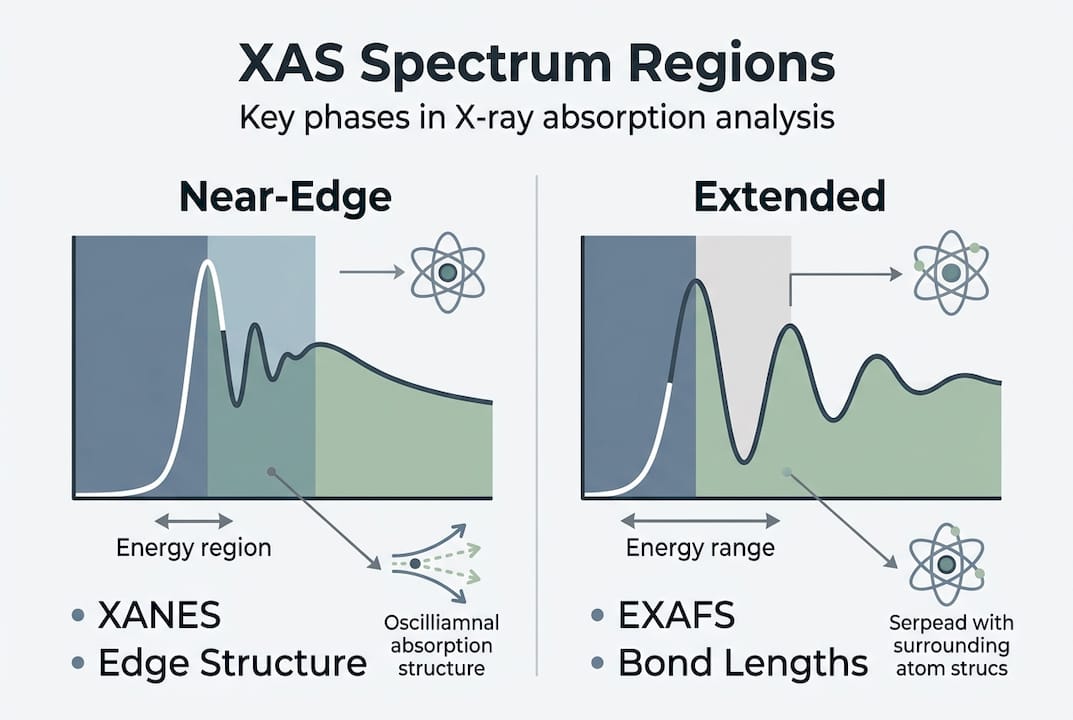
The XAS spectrum is divided into two distinct regions. The X-ray absorption near-edge structure (XANES) spans roughly 50 eV around the edge and encodes oxidation state and coordination geometry. The extended X-ray absorption fine structure (EXAFS) begins about 30 eV above the edge and provides interatomic distances and coordination numbers out to approximately 5 to 6 Å. Together, these two regions give you a complete picture of local atomic structure, even in amorphous or disordered materials where XRD produces no usable signal.
Compare that to the alternatives:
| Technique | What it measures | Primary applications |
|---|---|---|
| XAS | Local atomic structure, oxidation state, coordination | Amorphous phases, trace metals, operando studies |
| XRD | Long-range crystal structure, phase identification | Crystalline materials, phase quantification |
| XRF | Elemental composition | Bulk elemental mapping, quality control |
The key outputs that set XAS apart include:
- Oxidation state of a specific element within a complex mixture
- Interatomic bond lengths with precision around 0.02 Å
- Coordination number even in non-crystalline environments
- Local symmetry and geometry from XANES fingerprinting
- Speciation of trace elements at concentrations below 1%
“XANES is particularly powerful because it is sensitive to both the formal oxidation state and the local coordination geometry of the absorbing atom, making it indispensable for characterizing catalysts, metalloproteins, and battery materials where multiple chemical states coexist.” — XAS methodology literature
This combination of element specificity and structural sensitivity is what makes XAS irreplaceable in advanced materials characterization. No other benchtop or synchrotron technique simultaneously delivers oxidation state and local bonding information for a single element embedded in a complex matrix.
How XAS works: Modes, energies, and detection
With the basic principles clear, it is essential to understand the practical modalities and instrumentation choices that shape what XAS can reveal from your samples.
XAS requires a tunable X-ray source. Synchrotrons are the standard because they deliver high-flux, energy-tunable beams across a wide range. The energy regime determines which elements and sample environments are accessible: soft X-rays below 2 keV suit light elements like carbon, nitrogen, and oxygen but require vacuum conditions; tender X-rays from 2 to 6 keV cover elements like sulfur, phosphorus, and chlorine with some environmental flexibility; hard X-rays above 6 keV access heavier elements such as iron, copper, and platinum and work in air or through liquid cells.
Collecting a XAS spectrum follows a defined sequence:
- Align the sample at the beamline and calibrate the monochromator using a reference foil of the target element.
- Set the energy scan range, typically 200 eV below the edge to 1000 eV above, to capture both XANES and EXAFS regions.
- Record the incident beam intensity (I₀) using an upstream ionization chamber.
- Measure transmitted or emitted signal depending on your chosen detection mode.
- Normalize and background-subtract the raw data before extracting structural parameters.
Detection mode selection is critical and depends on sample concentration and physical form. Transmission mode uses ionization chambers to measure I₀ and the transmitted intensity (It), making it ideal for homogeneous samples at concentrations above roughly 1%. Fluorescence detection collects the characteristic X-ray emission at 90 degrees to the incident beam, making it the right choice for dilute or thin samples. Electron yield (TEY) detection is surface-sensitive, probing only the top few nanometers, which suits surface chemistry studies.

| Detection mode | Best sample type | Sensitivity | Depth probed |
|---|---|---|---|
| Transmission | Concentrated, homogeneous | Moderate | Bulk |
| Fluorescence | Dilute, thin films | High | Bulk to near-surface |
| Electron yield (TEY) | Surface layers | Very high | 2 to 10 nm |
Pro Tip: For trace metal samples or thin-film electrodes, fluorescence spectroscopy detection maximizes signal, but self-absorption artifacts can distort peak intensities at higher concentrations. Always validate your fluorescence data against a transmission measurement when sample concentration allows, and apply established correction algorithms when it does not.
Synchrotron tunability matters beyond just reaching the right energy. Precise edge scans require energy resolution better than 0.5 eV in most XANES work, and only synchrotron monochromators reliably deliver that. Laboratory-based XAS sources are emerging but remain limited in flux and energy range, making synchrotron access the practical standard for publication-grade data.
Interpreting XAS data: Case studies and benchmarks
Once spectra are acquired, the challenge becomes extracting meaningful information. Here is how researchers make practical sense of the data.
Reference standards anchor every reliable XAS analysis. Without them, you cannot distinguish real chemical changes from artifacts introduced by sample preparation or beam effects. Commonly used calibration benchmarks include:
- Copper foil: EXAFS fitting confirms a coordination number of approximately 12 and a Cu-Cu bond length near 2.5 Å, serving as a universal first-shell calibration standard.
- Iron tris-phenanthroline complexes: Fe(phen)₃ XANES shifts clearly distinguish low-spin states with Fe-N bonds at 1.97 Å from high-spin states at 2.08 Å, making this system a textbook benchmark for spin-state sensitivity.
- Iron oxides: Magnetite, hematite, and goethite each produce distinct XANES edge positions and white-line shapes, enabling oxidation state fingerprinting in environmental and catalytic samples.
- Platinum nanoparticles: Pt foil and PtO₂ references bracket the oxidation state range and help quantify Pt valence in fuel cell catalysts.
Two case studies illustrate how these benchmarks translate to real research outcomes. In battery cathode research, EXAFS fitting of nickel-rich layered oxides during charge-discharge cycling reveals systematic contraction and expansion of Ni-O bond lengths correlated with capacity fade. Teams use copper foil calibration at the start of every beamtime session to confirm their fitting protocol before applying it to the cathode data. In metalloprotein studies, XANES edge positions and pre-edge features of iron centers in hemoglobin variants are compared against iron porphyrin reference compounds to assign oxidation and spin states without requiring crystallographic data.
“Local structural information from XAS fills a critical gap in materials characterization: it provides atomic-level detail in systems where long-range order is absent or irrelevant to the property of interest.”
Pro Tip: Always collect reference spectra during the same beamtime session as your samples. Energy calibration can drift between sessions, and even a 0.5 eV shift in edge position can be misread as a change in oxidation state if references are not co-measured.
The combination of XANES fingerprinting and EXAFS fitting gives your team a quantitative, reproducible framework for tracking structural evolution across synthesis conditions, processing steps, or operational cycles.
Real-world applications: Pharmaceutical and materials breakthroughs
These empirical examples lead directly to higher-level applications. Here is how industries leverage XAS for critical innovation.
In pharmaceutical and biomedical research, XAS characterizes drug crystalline forms and protein-metal complexes with a level of specificity that standard spectroscopic tools cannot reach. Key application areas include:
- Drug solid-state characterization: XANES distinguishes different coordination environments of metal-containing active pharmaceutical ingredients, supporting polymorph identification and regulatory submissions.
- Metalloprotein and enzyme studies: Researchers use XAS to map the oxidation state and coordination of zinc, copper, and iron centers in target proteins, directly informing inhibitor design.
- Contrast agent development: Local structure around gadolinium or manganese centers in MRI contrast agents is validated by EXAFS to confirm coordination number and stability before in vivo testing.
In materials science and energy research, operando XAS has become a standard tool for studying battery cathodes and electrocatalysts under working conditions. Local structure changes in lithium-ion cathode materials during cycling are tracked in real time, revealing mechanisms of structural degradation that bulk XRD cannot resolve because the changes are local and disordered. In electrocatalysis, XAS speciation identifies the active oxidation state of transition metal sites during oxygen evolution or CO₂ reduction, guiding rational catalyst design.
“The ability to track local structural changes in operando conditions, without disrupting the active state of a material, has transformed how we understand battery degradation and catalytic activity at the atomic scale.” — Advanced material characterization literature
In aerospace and advanced engineering, XAS resolves oxidation and corrosion mechanisms in high-temperature alloys at the element-specific level, providing data that informs alloy composition and protective coating design. These cross-sector applications share a common theme: XAS delivers local structural truth where other methods deliver only averaged or compositional information.
Nuances and pitfalls: Advanced considerations in XAS
Real-world deployment makes it vital to be aware of technical nuances. Here is how to avoid the most common XAS mistakes and artifacts.
Even experienced teams encounter problems that compromise data quality or lead to incorrect structural conclusions. The most consequential pitfalls are:
- Beam damage: Prolonged X-ray exposure, especially in operando or cryogenic experiments, can reduce metal centers or alter coordination environments. Monitor for beam damage by collecting repeat scans and checking for spectral drift.
- Self-absorption in fluorescence mode: At higher concentrations, emitted fluorescence is reabsorbed before reaching the detector, suppressing peak intensities and distorting EXAFS amplitudes. Anti-resonance effects in soft X-ray fluorescence yield are particularly severe and require correction, especially when using high-energy-resolution fluorescence detection (HERFD-XAS).
- Sample thickness effects: Non-uniform or too-thick samples in transmission mode introduce “pinhole” artifacts that distort the edge jump and EXAFS oscillations.
- Oversimplified structural models: Fitting EXAFS with too few shells or ignoring multiple scattering paths produces bond lengths and coordination numbers that look precise but are physically wrong.
“XANES interpretation requires multiple scattering theory, not the single-scattering approximation sufficient for EXAFS. Teams that apply single-scattering models to near-edge data systematically misassign coordination geometry and symmetry.” — Nature Reviews Chemistry
Integrating theoretical calculations with experimental XAS data is no longer optional for complex systems. Ab initio codes such as FEFF, FDMNES, and WIEN2k generate simulated spectra that can be compared directly to experimental XANES, helping assign features and avoid overfitting. Machine learning approaches are increasingly applied to materials compliance methods that involve large spectral datasets, enabling rapid oxidation state mapping across hundreds of spectra without manual fitting.
Pro Tip: When possible, collect data in both transmission and fluorescence modes on the same sample. Comparing the two immediately flags self-absorption artifacts and thickness effects, saving you from building an entire analysis on compromised data.
Why most XAS interpretations fall short—and how to get it right
After addressing technical nuances, it is worth examining why conventional analysis so often produces misleading results in advanced characterization work.
The core problem is that XAS is an ensemble-averaged technique. A single spectrum reflects the average local environment of all absorbing atoms in the beam path. When a material contains multiple coexisting phases, surface-specific chemistry, or dynamic interfacial species, that average obscures exactly the information your team needs. Bulk measurements routinely wash out local chemical changes at interfaces, which is precisely where catalysis, corrosion, and electrochemical reactions happen.
What actually works is designing experiments to capture that heterogeneity rather than averaging it away. Operando methods, modulation-excitation XAS, and ab initio molecular dynamics (AIMD) ensembles all address the averaging problem by introducing time or perturbation as a variable, isolating the spectral response of active species from the static background. Coupling XAS with AI and ML models trained on reference databases accelerates the deconvolution of mixed-phase spectra without the bias introduced by manual model selection.
We consistently find that teams underestimate the importance of lab characterization strategies that pair XAS with complementary surface-sensitive techniques. Combining XAS with XPS or SIMS, for example, separates bulk and surface contributions rather than forcing one technique to answer both questions.
Pro Tip: Design your experiment to isolate both surface and bulk contributions from the start. Retrofitting surface sensitivity after data collection is rarely successful and often leads to ambiguous conclusions.
Unlock the full power of XAS in your research
Translating XAS principles into reliable, publication-grade results requires more than beamtime access. It requires expert experimental design, rigorous data processing, and interpretation grounded in complementary characterization.

At Materials Metric, we support research teams across pharmaceutical, biomedical, and advanced materials sectors with advanced material characterization solutions that integrate XAS with integrated chemical and microscopy approaches for complete structural insight. Our chemical and elemental analysis service is designed to fit your R&D pipeline, from initial feasibility through regulatory submission. We are here to function as an extension of your research team, delivering the precision and context your data demands.
Frequently asked questions
How does XAS differ from XRD and XRF?
XAS reveals local atomic structure and electronic state of specific elements, while XRD finds long-range crystal structure and XRF identifies bulk elemental composition. Unlike XRD, XAS works on amorphous and disordered materials because it does not require crystalline order, and unlike XRF, it provides bonding and oxidation state information beyond elemental identity.
What are the main limitations of XAS?
Key limitations include beam damage during long exposures, self-absorption distortions in fluorescence mode spectra, and the ensemble-averaging effect that obscures local dynamics in heterogeneous samples.
When should I use transmission vs fluorescence detection in XAS?
Use transmission for concentrated, homogeneous samples above roughly 1% target element concentration, and use fluorescence for dilute or thin-film samples, applying self-absorption corrections when concentration is elevated.
How precise are bond lengths and coordination numbers in XAS?
Typical EXAFS precision is within approximately 0.02 Å for bond lengths, as confirmed by benchmarks such as Cu foil at 2.5 Å, while coordination numbers carry roughly 10 to 20% uncertainty depending on data quality and model complexity.
What computational methods help interpret complex XAS spectra?
Ab initio codes, machine learning classifiers, and sum rules for quantification are the primary tools for interpreting complex XAS data, particularly when multiple scattering contributions dominate the XANES region or when mixed-phase spectra require deconvolution.



