TL;DR:
- Selecting a highly validated, compliant chemical testing lab is crucial for regulatory success.
- Core analytical methods include ICP-MS, ICP-OES, GC/MS, XRF, XRD, and AA, each suited for specific applications.
- Proper validation, transparency, and expert interpretation ensure data supports product quality and regulatory submissions.
Not all chemical testing labs are created equal, and assuming otherwise can put your product, your compliance record, and your regulatory submissions at serious risk. The method a lab selects, the rigor of its validation protocols, and the depth of its technical expertise determine whether your data will hold up under FDA scrutiny or collapse under audit pressure. Across biomedical, pharmaceutical, aerospace, and manufacturing sectors, the stakes of poor lab selection are measurable in recalls, failed submissions, and lost development time. This guide covers the core functions of advanced chemical testing labs, the major analytical methodologies and their trade-offs, what validated compliance really looks like, and how to translate lab data into actionable product and quality outcomes.
Table of Contents
- Understanding chemical testing labs: Core roles and value
- Key chemical analysis methodologies: Techniques, strengths, and trade-offs
- Compliance and validation: Meeting ICH, FDA, and Eurachem standards
- Applying lab data: Accelerating product development and controlling quality
- Perspective: Why not all chemical testing labs deliver equal value
- Finding advanced solutions with Materials Metric
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Lab capabilities vary | Choose labs with proven expertise in your materials and industry standards for best results. |
| Method selection matters | Advanced tests like ICP-MS or GC/MS should fit the specific challenge, not a one-size-fits-all approach. |
| Compliance is non-negotiable | Demand documented method validation and certifications to meet regulatory expectations. |
| Data drives action | Leverage lab results for faster product development, troubleshooting, and quality control. |
Understanding chemical testing labs: Core roles and value
A chemical testing lab is far more than a facility that runs samples and returns numbers. At its core, it provides analytical testing services that generate scientifically defensible data about a material’s composition, purity, contamination profile, and performance characteristics. For regulated industries, that data is the foundation of every compliance submission, quality release decision, and failure investigation.
Understanding what chemical analysis actually encompasses helps set the right expectations. It includes elemental composition profiling, trace contaminant identification, molecular structure verification, and quantitative purity assessment. Each of these functions serves a distinct purpose depending on the industry.
Here is how the role of a chemical testing lab shifts by sector:
- Biomedical and pharmaceutical: Labs support raw material qualification, extractables and leachables studies, drug substance purity, and impurity profiling per ICH guidelines. Batch consistency evaluation is critical for demonstrating manufacturing control.
- Aerospace: Material composition verification, alloy certification, and failure analysis of components under stress or environmental exposure are common needs.
- Manufacturing: Quality control testing, supplier qualification, and incoming material verification prevent defective inputs from entering production lines.
- Energy and advanced materials: Surface chemistry, corrosion product identification, and coating integrity assessments support long service life and safety.
Regulatory compliance is a central driver across all of these. Validation per ICH/FDA/Eurachem guides is essential for any method used in a regulated context, meaning labs must demonstrate that their procedures are accurate, reproducible, and fit for the intended purpose. A lab that cannot produce documented validation data is not a viable partner for regulated submissions.
Beyond compliance, advanced labs contribute to product R&D by identifying failure modes early, characterizing novel materials, and providing data that informs formulation decisions. Treating a chemical testing lab as a commodity service is a strategic error. The right lab functions as an extension of your technical team, providing interpretation alongside raw data.
Key chemical analysis methodologies: Techniques, strengths, and trade-offs
With the lab’s role established, it is essential to understand the core testing methods that drive advanced analysis. Method selection is not arbitrary. It depends on the analyte type, required sensitivity, matrix complexity, and regulatory expectations.
Here are the primary techniques used in advanced chemical testing:
- ICP-MS (Inductively Coupled Plasma Mass Spectrometry): Delivers parts-per-trillion (ppt) sensitivity for trace metals. Best for ultra-clean matrices and low-level elemental analysis in pharma and biomedical applications.
- ICP-OES (Inductively Coupled Plasma Optical Emission Spectrometry): Operates at parts-per-billion (ppb) sensitivity with higher tolerance for complex, high-dissolved-solids matrices. Preferred when sample matrices are challenging.
- GC/MS (Gas Chromatography Mass Spectrometry): The standard for volatile and semi-volatile organic compound identification and quantification. Used extensively for residual solvents, nitrosamines, and organic impurities.
- XRF (X-ray Fluorescence): Rapid, non-destructive elemental analysis across a broad elemental range. Ideal for alloy verification and surface composition screening.
- XRD (X-ray Diffraction): Identifies crystalline phases and polymorphic forms. Critical in pharmaceutical solid-state characterization.
- AA (Atomic Absorption Spectrometry): Cost-effective for single-element analysis in routine quality control applications.
| Method | Sensitivity | Best for | Key limitation |
|---|---|---|---|
| ICP-MS | ppt | Ultra-trace metals, pharma | Polyatomic interferences |
| ICP-OES | ppb | High-matrix metals analysis | Lower sensitivity than ICP-MS |
| GC/MS | ppt to ppb | Organics, solvents, volatiles | Requires volatile analytes |
| XRF | ppm | Alloy screening, surfaces | Poor for light elements/trace |
| XRD | Phase level | Crystalline structure | Not for amorphous materials |
| AA | ppb | Single-element routine QC | Slow for multi-element |
Selecting the wrong method introduces systematic errors that no amount of statistical treatment can correct. ICP-MS is prone to polyatomic interferences requiring correction strategies, while ICP-OES handles high total dissolved solids matrices more reliably. XRF is fast and non-destructive but performs poorly for light elements and very low concentrations.
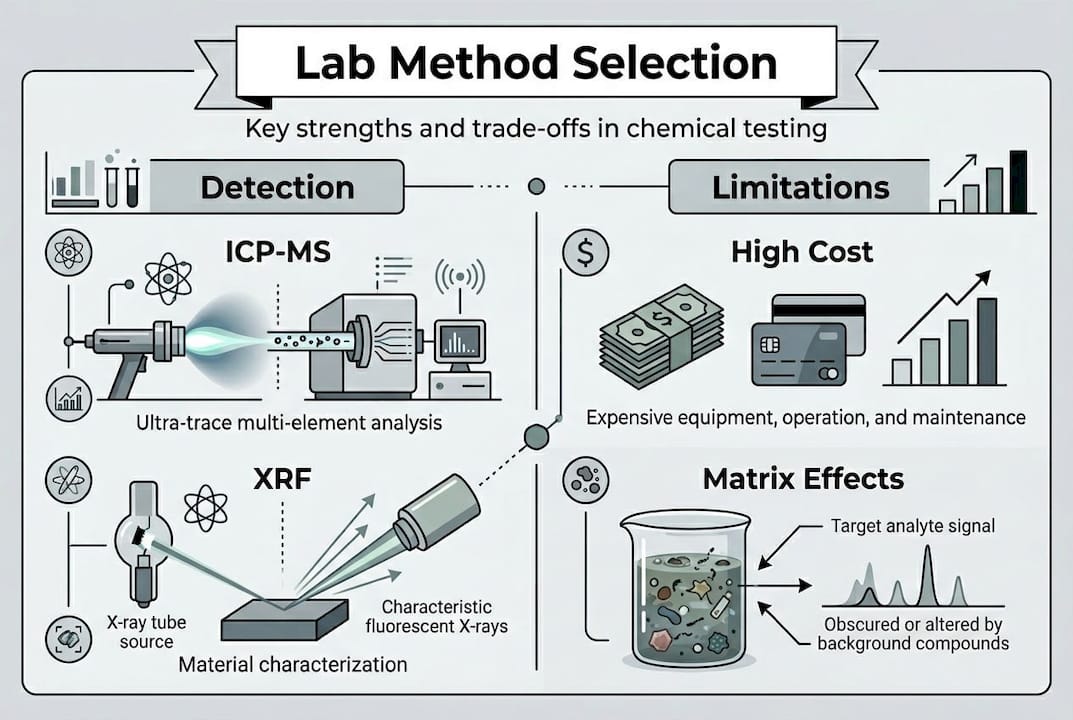
Pro Tip: Always discuss your sample matrix with the lab before method selection. A high-salt biological matrix that works well with ICP-OES may suppress signals in ICP-MS without proper internal standard correction, leading to falsely low results. Experienced labs will flag this upfront and recommend appropriate method development and validation steps.
Compliance and validation: Meeting ICH, FDA, and Eurachem standards
Once testing methods are selected, understanding validation and compliance is paramount, especially in highly regulated industries. Method validation is the formal process of demonstrating that an analytical procedure consistently produces results that are accurate, precise, and fit for their intended regulatory purpose.
Three major frameworks govern this process:
- ICH (International Council for Harmonisation): Sets globally recognized guidelines for pharmaceutical method validation, including Q2(R1) for analytical procedures.
- FDA (Food and Drug Administration): Requires validated methods for any data submitted in drug applications, device submissions, or manufacturing records.
- Eurachem: Provides practical guidance for laboratories across sectors on method validation and measurement uncertainty.
Validation per ICH/FDA/Eurachem guides is essential for compliance, and the parameters that must be documented include accuracy, precision, linearity, specificity, limit of detection (LOD), limit of quantitation (LOQ), and robustness.

| Validation parameter | Definition | Relevant industries |
|---|---|---|
| Accuracy | Closeness to true value | Pharma, biomedical, food |
| Precision | Reproducibility of results | All regulated sectors |
| Linearity | Response across concentration range | Pharma, environmental |
| Specificity | Ability to distinguish analyte | Pharma, biomedical |
| LOD/LOQ | Lowest detectable/quantifiable level | All sectors |
| Robustness | Stability under variable conditions | Pharma, manufacturing |
Understanding the importance of analytical testing in this context means recognizing that a method that has not been formally validated cannot be used to support a regulatory submission, regardless of how technically sophisticated the instrumentation is. Validation documentation is what transforms raw instrument output into legally defensible data.
When evaluating a lab’s compliance posture, request their full method validation packages, certificates of analysis with traceability statements, and evidence of participation in proficiency testing programs. Labs that offer method validation services as a documented deliverable, not just an internal process, are better positioned to support your audit readiness.
A lab that hesitates to share validation data is a red flag. Transparency in documentation is a non-negotiable requirement for any partner supporting regulated product development.
Applying lab data: Accelerating product development and controlling quality
With a solid grasp of labs’ technical and compliance foundations, the next step is understanding how to apply their data directly for tangible business and technical outcomes. Raw analytical results only create value when they are correctly interpreted and acted upon.
Here is a practical workflow from sample submission to actionable outcome:
- Define the analytical question clearly. Specify whether you need composition profiling, contaminant identification, or comparative batch analysis before submitting samples.
- Select the appropriate method with the lab. Discuss matrix effects, required sensitivity, and regulatory context upfront to avoid method mismatch.
- Submit samples with full context. Provide manufacturing history, storage conditions, and any known interferences. This directly affects data quality.
- Review the report with a technical contact. Do not accept a data package without a technical discussion. Outliers, matrix effects, and borderline results need expert interpretation.
- Integrate findings into QA or development decisions. Use the data to support batch release, inform formulation changes, or document compliance evidence for audits.
Real-world applications include contaminant screening for high-potency active pharmaceutical ingredients (HPAPIs) and nitrosamine impurities, both of which require custom analytical methods validated to specific regulatory thresholds.
Choosing the right lab is not just a procurement decision. It is a regulatory strategy. The lab you select will directly influence whether your data package survives an FDA inspection or requires costly remediation.
Pro Tip: Before your next audit, request a mock data review with your lab’s technical team. Walk through a recent report and ask them to explain every flagged result, every uncertainty value, and every deviation note. If they cannot do this fluently, your audit readiness is at risk.
Perspective: Why not all chemical testing labs deliver equal value
The uncomfortable truth is that many labs meet minimum technical specifications on paper but vary dramatically in what they actually deliver. Accreditation certificates and instrument lists tell you what a lab can do. They do not tell you how well the team interprets ambiguous data, how quickly they respond when a result is unexpected, or how thoroughly they document edge cases that matter during an FDA audit.
Sophisticated clients in pharma and biomedical sectors have learned this through experience. They do not evaluate labs on turnaround time and price alone. They evaluate them on the quality of technical dialogue, willingness to customize methods for novel matrices, and depth of validation documentation provided as a standard deliverable.
The labs that consistently deliver value are the ones that engage with your problem before the sample is even submitted. They ask questions. They flag potential interferences. They recommend method adjustments proactively. Understanding the impact of analytical testing on compliance outcomes makes it clear why this level of engagement is not optional. It is the difference between data that supports your submission and data that creates more questions than it answers.
Finding advanced solutions with Materials Metric
Having learned how labs drive compliance and innovation, the logical next step is connecting with a partner that operates at this level of technical depth and regulatory rigor.

At Materials Metric, we provide analytical testing compliance support across biomedical, pharmaceutical, aerospace, and manufacturing sectors. Our capabilities span integrated chemical and microscopy characterization through to chemical and elemental characterization using ICP-MS, ICP-OES, GC/MS, XRF, and more. We do not just run samples. We interpret results, support audit preparation, and develop custom methods tailored to your regulatory context. Reach out with your project specifics and let us help you build a data package that holds up.
Frequently asked questions
What certifications should a top chemical testing lab have?
A top lab should be ISO 17025 accredited and demonstrate validation per ICH/FDA/Eurachem guidelines for any method used in regulated submissions. Accreditation scope and validation documentation should be available on request.
How do labs handle trace metals vs. organic contaminants?
Trace metals are analyzed using ICP-MS or ICP-OES depending on matrix complexity and required sensitivity, while organic contaminants require GC/MS or LC/MS for high-resolution identification and quantification. Method selection depends on the regulatory threshold and sample type.
What makes XRF analysis unique for material testing?
XRF provides rapid, non-destructive elemental screening across a broad range of materials without sample destruction, making it ideal for alloy certification and surface analysis. Its limitation is reduced sensitivity for light elements and very low trace concentrations.
Why is method validation critical in regulated industries?
Method validation establishes that a test procedure produces accurate, legally defensible results that can withstand regulatory scrutiny during submissions and inspections. Without it, even technically sound data cannot be used to support compliance claims.
Recommended
- Analytical testing explained: methods, compliance, and impact
- Laboratory assays: Methods, validation, and compliance
- What is chemical analysis ? Methods, Types & Applications
- Importance of Analytical Testing | Materials Metric | Chemistry
- Industry Standards for Spray Booth Films: Ensuring Compliance and Superior Finishes



