TL;DR:
- Wet chemistry remains a trusted, regulation-approved framework critical for compliance in regulated industries.
- Classical techniques like titrimetric and gravimetric analysis provide direct, accurate, and traceable results.
- Industries like pharma, aerospace, energy, and biomedical rely on wet chemistry for raw material verification and quality control.
Rapid advances in spectroscopy, mass spectrometry, and automated instrumentation have led some product development teams to assume that classical wet chemistry belongs in the past. That assumption carries real risk. For regulated industries, including pharmaceutical manufacturers, biomedical device companies, aerospace suppliers, and energy sector material engineers, wet chemistry is not a legacy technique. It is a living, validated framework that regulators and auditors trust precisely because of its quantitative rigor and established traceability. This article clarifies what wet chemistry includes, how its core methods work, where specific industries rely on it most, and why compliance programs without it face gaps that instrumentation alone cannot fill.
Table of Contents
- What is wet chemistry? Definitions and core concepts
- Titrimetric and gravimetric analysis: Methods and best-fit applications
- Wet chemistry in industry: Biomedical, pharma, aerospace, and energy use cases
- Regulatory acceptance and compliance standards
- Why classic wet chemistry endures: A material scientist’s view
- Access advanced wet chemistry and compliance support
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Foundational approach | Wet chemistry remains the backbone of high-accuracy analysis in advanced industries despite digital advancements. |
| Compliance assurance | Major standards like USP, ASTM, ISO, and EPA endorse wet chemistry for audit-ready material verification. |
| Versatile methodology | From pharmaceuticals to energy, wet chemistry adapts to complex sample needs for reliable results. |
| Strategic application | Choosing the right wet chemistry method optimizes quality, risk reduction, and regulatory alignment. |
What is wet chemistry? Definitions and core concepts
Wet chemistry refers to a family of classical analytical techniques performed in liquid-phase environments, using chemical reactions to identify, quantify, or characterize substances. The term “wet” distinguishes these solution-based methods from dry instrumental techniques such as X-ray fluorescence or infrared spectroscopy. These methods have been refined over more than a century and remain central to materials science because they deliver direct, reproducible quantitative results with minimal matrix dependency.
Our wet chemistry testing services cover the principal technique categories that practitioners in compliance-heavy industries need most:
- Titrimetric analysis: Measures analyte concentration through precisely calibrated volumetric reactions with a standard solution until an endpoint is reached
- Gravimetric analysis: Determines composition or purity by measuring the mass of a chemically isolated component after precipitation and drying
- pH and conductivity measurement: Evaluates ionic concentration and solution behavior in material matrices
- Basic chemical assays: Functional group identification, solubility testing, and reactive species quantitation
- Moisture and ash content determination: Directly relevant to pharmaceutical formulation and raw material acceptance
As defined in our classical analytical methods documentation, wet chemistry encompasses classical analytical techniques including titrimetric, gravimetric, and basic chemical assays for quantitative and qualitative evaluation of chemical composition, purity, reactivity, moisture, and solubility.
Wet chemistry is not a workaround for labs that lack instrumentation. It is often the most direct route to a defensible, audit-ready result — particularly in matrices where spectroscopic interference or sample preparation artifacts can compromise instrument-based readings.
The practical reason wet chemistry remains valued alongside modern tools is specificity. When a regulator asks how much active pharmaceutical ingredient is present in a formulation, or whether a composite material meets a specific purity threshold, a titrimetric result tied to a USP or ASTM reference method carries weight that a software-derived spectral deconvolution simply may not.
Titrimetric and gravimetric analysis: Methods and best-fit applications
Having set the stage with foundational definitions, this section dives into key methods you will encounter across materials testing projects.
Titrimetric analysis works by introducing a standard reagent of known concentration into a sample solution until the reaction reaches a defined endpoint, often signaled by an indicator color change or an electrochemical shift. The volume of reagent consumed relates directly to the analyte concentration. This approach is used to quantify acids, bases, oxidizing agents, reducing agents, and metal ions.
Gravimetric methods follow a different logic: isolate the analyte as a chemically stable precipitate, dry it under controlled conditions, and weigh it with calibrated instrumentation. The mass obtained is proportional to the concentration or content of the target species. No calibration curve is needed, which is one reason gravimetric results are often used as reference values for instrument calibration.
Comparison: titrimetry vs. gravimetry in materials testing
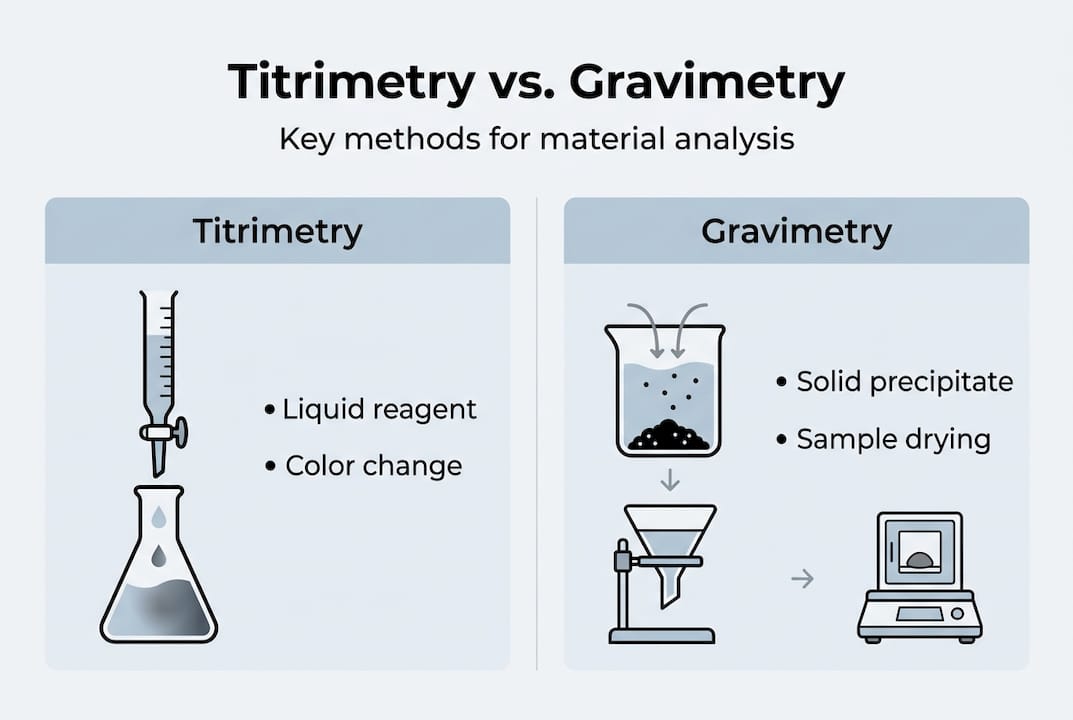
| Factor | Titrimetric analysis | Gravimetric analysis |
|---|---|---|
| Measurement basis | Reaction volume | Isolated mass |
| Speed | Moderate to fast | Slower (drying step required) |
| Accuracy | High | Very high |
| Best for | Acids, bases, metal ions | Purity, moisture, ash content |
| Regulatory fit | USP, ASTM, ISO | USP, ASTM |
| Typical sector | Pharma, energy | Aerospace, pharma, environmental |
Key applications by sector:
- Pharmaceutical: Titrimetric assays confirm active ingredient content in dosage forms; gravimetric loss-on-drying establishes moisture specifications
- Aerospace composites: Gravimetric methods confirm resin content and assess fiber-to-matrix ratios after chemical extraction
- Energy materials: Titrimetric methods quantify acid number in lubricants, electrolyte concentration in batteries, and sulfur content in fuels
As documented in regulatory standards, key methodologies include titrimetric analysis for quantifying acids, bases, oxidizers, and metal ions via volumetric reactions aligned with ASTM, USP, and ISO; gravimetric methods measure mass-based purity and composition.
Pro Tip: The most common source of error in titrimetric analysis is a sluggish or ambiguous endpoint detection. Always pre-calibrate your indicator against a certified reference material and run at least three replicate titrations before reporting. For gravimetric work, verify your drying temperature and equilibration time against the relevant standard protocol, since under-drying inflates moisture values while over-drying can decompose certain materials.
Wet chemistry in industry: Biomedical, pharma, aerospace, and energy use cases
With an understanding of main methods, now see how sectors use these tools strategically.
Wet chemistry does not serve all industries the same way. The techniques are the same; the compliance stakes and the matrices differ significantly.
Biomedical and pharmaceutical applications
In pharmaceutical manufacturing, every raw material arriving at the receiving dock must be verified against a certificate of analysis. Wet chemistry provides the primary assay method for most compendial raw materials under USP and EP monographs. Beyond raw material acceptance, it supports:
- Formulation quality control during development and scale-up
- Drug substance quantitation in biological fluids using hybrid approaches such as ion-pair reversed-phase liquid chromatography mass spectrometry
- Oligonucleotide quantitation where classical UV methods are insufficient
- Extractables and leachables testing for container closure systems and medical devices
For biomedical device manufacturers, wet chemistry is integral to verifying biocompatibility-relevant chemical properties before ISO 10993 biological evaluation.
Aerospace and energy sector applications
Aerospace material engineers use wet chemistry to investigate composite failure under chemical loads, verify surface treatment bath concentrations, and document pre-treatment compliance. Energy sector applications focus on materials used in extreme environments, where chemical degradation is both a safety and a performance issue.

| Industry | Wet chemistry method | Typical application |
|---|---|---|
| Pharmaceutical | Titrimetric assay | Active ingredient content |
| Biomedical devices | Extraction and assay | Leachable quantitation |
| Aerospace | Gravimetric extraction | Resin content in composites |
| Energy | Titrimetric | Acid number, electrolyte strength |
| Environmental | Wet extraction (WET test) | Leachability and soil compliance |
As applied in biomedical and pharma contexts, wet chemistry supports formulation QC, raw material verification, oligonucleotide quantitation, and drug determination in fluids; aerospace and energy applications include composite failure analysis and materials compliance testing. For a broader view of how these capabilities integrate into your testing program, our chemical testing labs compliance resource outlines how classical and advanced methods are coordinated effectively.
Wet chemistry remains foundational for advanced materials characterization across regulated sectors because it produces data that holds up in the most demanding audit and investigation settings.
Regulatory acceptance and compliance standards
These industry applications underscore the stakes. Next, understand why regulators trust wet chemistry above all.
Regulatory agencies do not accept methods arbitrarily. USP, ASTM, ISO, and EPA each maintain codified wet chemistry methods because these techniques have demonstrated long-term interlaboratory reproducibility and are difficult to falsify without leaving a paper trail. That traceability is exactly what auditors value.
Key regulatory frameworks and their wet chemistry requirements:
- USP: Monograph-specific titrimetric and gravimetric assays for pharmaceutical ingredients; loss-on-drying, residue-on-ignition
- ASTM: Standardized test methods for metals, composites, lubricants, and environmental samples
- ISO: Laboratory method validation requirements including those for compliance insights relevant to quality management systems
- EPA Part 136: Methods for water and wastewater analysis using wet chemistry
- WET test (Waste Extraction Test): Governs leachability testing for materials that may contact soil or groundwater
As referenced in our regulatory documentation, regulatory compliance aligns with USP, ASTM, ISO, and EPA methods including Part 136 and the Waste Extraction Test for leachability, making wet chemistry essential for pharma QC and environmental monitoring.
For audit defense, wet chemistry provides something instrumental data often cannot: a direct, first-principles result tied to a specific reference method. When a regulatory investigator questions a test result, the ability to demonstrate that the method followed USP 541 or ASTM D1475 precisely is a substantive defense.
Wet chemistry is the gold standard for high-accuracy, compliance-critical projects where traceable, method-specific results are required by regulators and legal stakeholders alike.
Pro Tip: When selecting an external testing laboratory for compliance work, request their specific standard operating procedures for the wet chemistry methods relevant to your project. A qualified GLP-compliant lab should be able to show you the exact method version, calibration records, and analyst competency documentation without hesitation. If that documentation is not immediately available, that is a signal to look elsewhere.
Why classic wet chemistry endures: A material scientist’s view
We work at the intersection of emerging instrumentation and established method frameworks every day, and one pattern is clear: laboratories that have abandoned wet chemistry in favor of purely instrumental workflows tend to encounter the most difficulty during regulatory audits and high-stakes failure investigations.
Instrumental methods are fast and information-rich, but they carry an inherent vulnerability: the result is only as reliable as the method validation that supports it in your specific matrix. When a novel material or unusual sample challenges those validated conditions, the instrument may not flag the problem. A well-executed titrimetric or gravimetric assay, by contrast, involves direct chemical interaction that is hard to misinterpret.
We have seen methods of chemical analysis serve as decisive arbiters in disputed product quality investigations precisely because classical results are method-transparent and verifiable by any competent analyst. The foundational role of wet chemistry in advanced materials characterization persists because reliability in complex matrices and regulatory acceptance cannot be replicated by speed alone. The most effective programs we support are those that use wet chemistry as a confirmation layer alongside modern instrumentation, not as a backup plan, but as a deliberate quality checkpoint built into the testing strategy from day one.
Access advanced wet chemistry and compliance support
If your product development or compliance program requires dependable wet chemistry data, the details of your testing strategy matter more than the volume of tests you run.

At Materials Metric, our analytical testing services cover the full spectrum from classical wet chemistry to advanced instrumentation, all coordinated under ISO 9001:2015 and GLP-aligned protocols. Whether you need chemical and elemental characterization for a novel material or full-scope compliance documentation for a regulated submission, our team functions as an extension of your R&D and quality organization. Explore our wet chemistry laboratory services to see how we can support your next development phase or regulatory filing with precision and confidence.
Frequently asked questions
What are the main advantages of wet chemistry over instrumental analysis?
Wet chemistry offers unmatched accuracy, regulatory acceptance, and reliable performance in complex sample matrices, making it the preferred method for compliance-critical and high-stakes material analysis programs.
When should I use gravimetric analysis in materials testing?
Gravimetric analysis is the right choice when precise, first-principles measurement of composition or purity is required, such as during raw material qualification, resin content verification, or mass-based purity determination in failure investigations.
Which regulatory methods commonly mandate wet chemistry?
USP, ASTM, ISO, and EPA all maintain codified wet chemistry procedures; USP, ASTM, ISO, EPA methods including EPA Part 136 and the Waste Extraction Test specifically require wet chemistry for pharmaceutical, environmental, and materials manufacturing compliance.
Is wet chemistry suitable for complex or novel materials?
Yes, wet chemistry frequently outperforms instrumental approaches in challenging or heterogeneous matrices because direct chemical reactions are less susceptible to matrix interference than spectroscopic or spectrometric signal acquisition.



