Understanding DSC Analysis: Your Essential Guide to Thermal Material Characterization
DSC analysis is a thermal analysis technique that measures the heat flow into or out of a sample compared to a reference as both are heated, cooled, or held at a constant temperature. This powerful method reveals critical information about material properties and transitions.
Quick Answer: What DSC Analysis Tells You
- Glass Transition Temperature (Tg) – When amorphous materials soften
- Melting Point (Tm) – Temperature at which crystalline materials melt
- Crystallization Temperature (Tc) – When materials form ordered structures
- Enthalpy Changes (ΔH) – Energy absorbed or released during transitions
- Heat Capacity (Cp) – Amount of heat needed to change temperature
- Purity and Stability – Quality metrics for pharmaceuticals and chemicals
- Curing and Reaction Kinetics – How materials polymerize or react over time
Whether you’re developing a new polymer formulation, validating pharmaceutical stability, optimizing food processing conditions, or characterizing advanced nanomaterials, DSC provides the thermal fingerprint you need to understand how your material behaves under heat.
Why DSC matters for your work: In the aerospace, biomedical, pharmaceutical, energy, and manufacturing sectors, thermal properties directly impact product performance, safety, and regulatory compliance. A material that softens at the wrong temperature can lead to catastrophic failure. A polymer with unexpected crystallinity can derail your production line. DSC helps you catch these issues early—before they become expensive problems.
The technique works by placing a small sample (typically 1-20 mg) in a pan alongside an empty reference pan. Both are heated or cooled in a controlled furnace, and the instrument measures the difference in heat flow needed to keep them at the same temperature. When your sample undergoes a thermal event—like melting, crystallizing, or changing from glassy to rubbery—the heat flow changes, creating peaks and shifts in the resulting thermogram.
From simple quality checks to complex kinetic studies, DSC delivers actionable data. It can tell you if your polymer batch has the right degree of cure, if your pharmaceutical compound contains unexpected polymorphs, or if your food ingredient will maintain stability during processing. The temperature range typically spans from -100°C to 700°C, with heating rates adjustable from 0.02 to 300°C/min, giving you flexibility to match real-world conditions or accelerate testing.
I’m Rupak Dua, Ph.D., Founder and CEO of Materials Metric, with deep expertise in materials characterization and validation strategy for regulated industries. Throughout my career leading polymer and materials research programs, I’ve relied on DSC analysis to guide formulation decisions, troubleshoot failures, and support regulatory submissions across medical devices, dental technologies, and advanced materials development.
Let’s explore how DSC works, what it can measure, and how to get the most reliable data from your experiments.
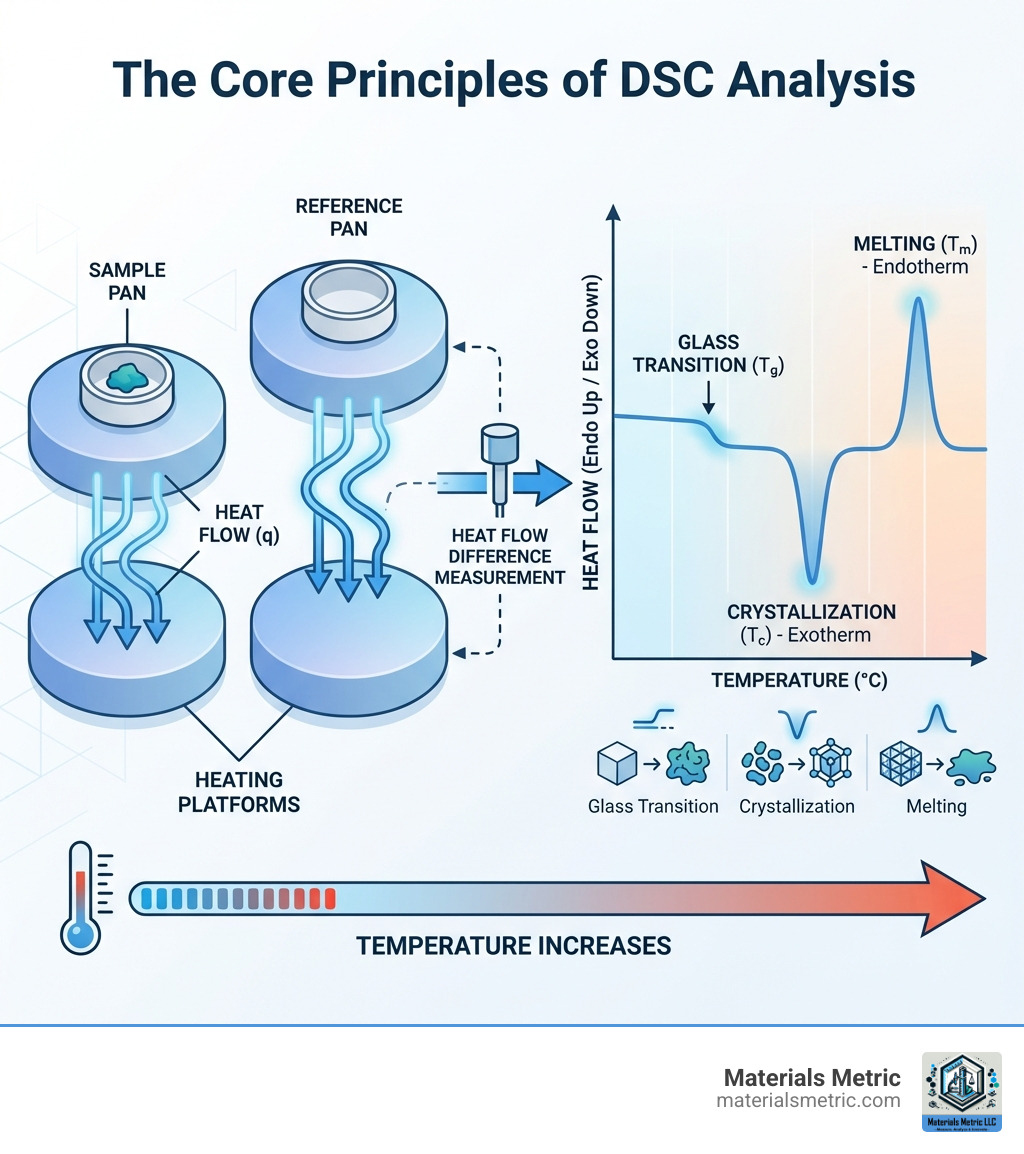
The Core Principles: How DSC Works
At its heart, Differential Scanning Calorimetry (DSC) measures the difference in heat flow between a sample and an empty reference pan as they are subjected to a controlled temperature program. This differential measurement is what makes DSC so powerful for detecting subtle thermal events.
The instrument contains two identical furnaces, one for the sample and one for the reference. As both are heated or cooled at a precise rate, the DSC works to keep their temperatures identical. When the sample undergoes a thermal event, the instrument must adjust the power supplied to the sample pan to maintain this temperature match. This power adjustment is the measured heat flow.
- Endothermic processes absorb heat. Events like melting, vaporization, or a glass transition require the DSC to supply more heat to the sample, creating an upward peak on the thermogram.
- Exothermic processes release heat. Events like crystallization, curing, or decomposition require the DSC to supply less heat, creating a downward peak.
Beyond peaks, DSC analysis also detects changes in a material’s heat capacity (Cp)—its ability to store heat. This appears as a shift in the curve’s baseline, most notably during a glass transition.
DSC instruments operate over a wide temperature range (typically -100°C to 700°C) with flexible heating rates (0.02 to 300°C/min) and cooling rates (0.02 to 50°C/min). This allows for simulating real-world conditions or accelerating tests on small samples (1-20 mg).
Fundamental Principles of DSC Analysis
The core of DSC analysis is its differential measurement. By comparing the sample to an inert reference, the instrument isolates the material’s intrinsic thermal behavior, canceling out instrumental effects. This relies on the principles of calorimetry and thermal equilibrium.
For accurate results, baseline stability is critical. A flat baseline is necessary to detect weak transitions and perform quantitative analysis. Early instruments struggled with baseline drift, but modern designs have largely overcome this. As noted in scientific literature (Thermochimica Acta, 198385073-4)), conventional DSC performance can be affected by non-ideal conditions. To correct for this, advanced calibration methods like Tzero calibration are used to account for thermal resistance and capacitance imbalances, leading to more accurate heat flow data.
Key Features of DSC Instruments
Modern DSC instruments fall into two main categories: Heat-Flux DSC and Power-Compensated DSC. Both measure differential heat flow but use different mechanisms to achieve it.
Here’s a summary of the key features:

| Feature | Heat-Flux DSC | Power-Compensated DSC |
|---|---|---|
| Measurement Principle | Measures temperature difference (ΔT) between sample and reference, then calculates heat flow. Both share a single furnace block. | Supplies differential power to separate heaters to maintain zero ΔT between sample and reference, directly measuring power difference. |
| Sensitivity | Generally good for strong transitions; can be limited for subtle events. | Often higher sensitivity, especially for weak transitions and heat capacity measurements. |
| Temperature Range | Wide range, typically -100 to 700 °C. | Wide range, typically -100 to 700 °C. |
| Heating/Cooling Rates | Good range (0.02 to 300 °C/min heating, 0.02 to 50 °C/min cooling). | Can achieve higher heating rates, supporting advanced techniques like Fast-Scan DSC. |
| Baseline Stability | Can be more prone to baseline drift and curvature. | Generally flatter, more stable baselines due to active power compensation. |
| Response Time | Slower response due to indirect heat flow measurement. | Faster response due to direct power adjustment. |
| Typical Applications | Melting, crystallization, Tg, purity, OIT. | Melting, crystallization, Tg (especially weak), heat capacity, kinetics, modulated DSC. |
While Heat-Flux DSCs are robust and common, Power-Compensated DSCs often provide superior precision for heat capacity and kinetic studies due to their faster response and more stable baselines. The choice depends on the specific application and required level of detail.
Decoding the Thermogram: Interpreting DSC Curves
The output of a DSC analysis is a thermogram, a plot of heat flow versus temperature that serves as a material’s “thermal fingerprint.” Interpreting this curve involves identifying several key features:
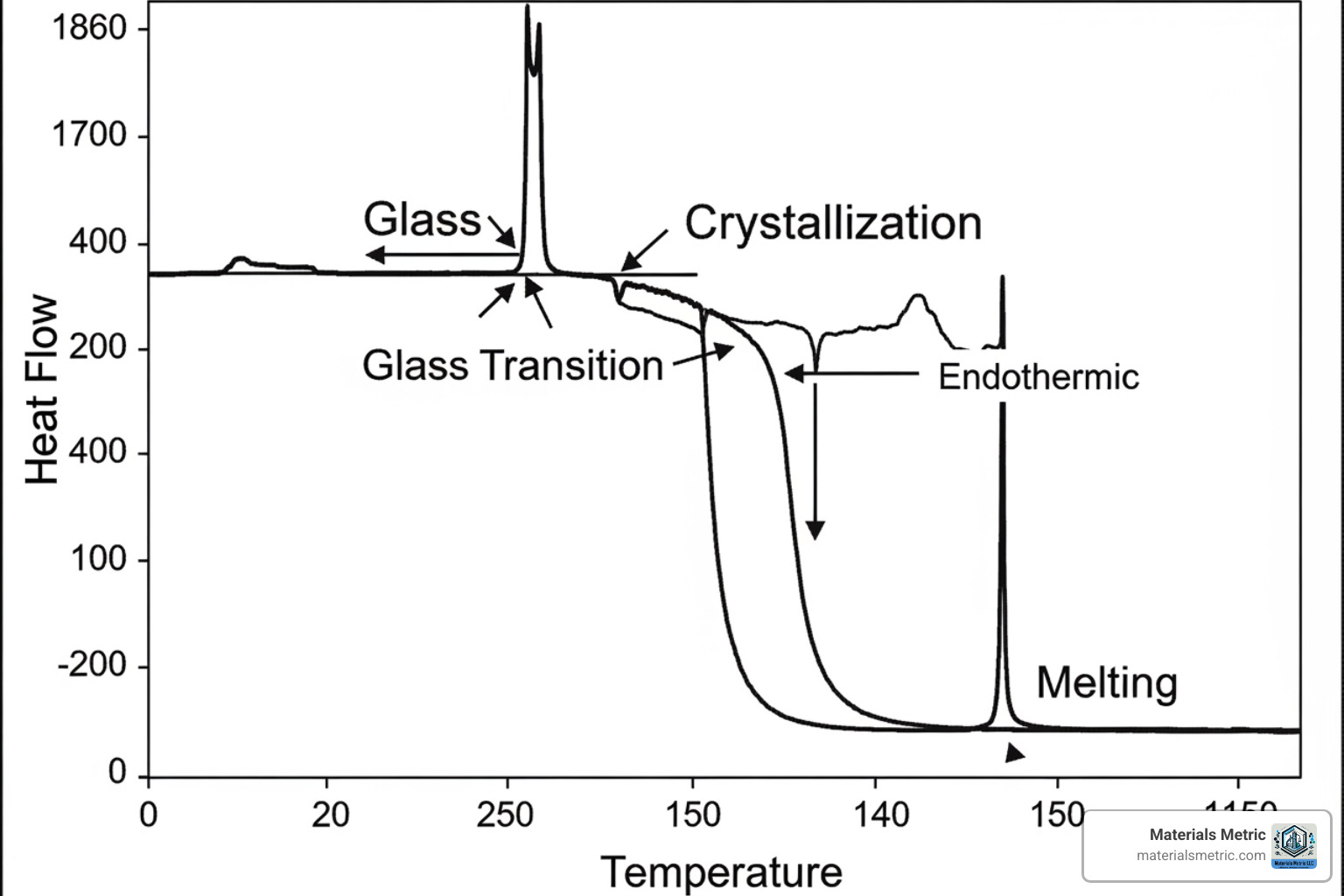
- Baseline: The flat portion of the curve where no thermal events occur. Its position relates to the material’s heat capacity.
- Baseline Shifts: A step-like change indicating a glass transition (Tg), where an amorphous material goes from a rigid to a rubbery state.
- Peaks: These represent enthalpy changes.
- Endothermic peaks (upward) show heat absorption, such as melting (Tm).
- Exothermic peaks (downward) show heat release, such as crystallization (Tc) or curing.
- Onset and Peak Temperatures: The start and maximum point of a transition, respectively. These are used to define characteristic temperatures like Tm.
- Enthalpy (ΔH): The area under a peak, which quantifies the energy absorbed or released during a transition.
Key Thermal Events Detected by DSC
DSC analysis can identify a wide range of thermal events:
- Glass Transition (Tg): A step change in the baseline unique to amorphous materials, marking the transition from a glassy to a rubbery state. It’s a change in heat capacity, not a true phase change.
- Crystallization (Tc): An exothermic peak that appears as a material’s molecules arrange into an ordered structure upon cooling or heating.
- Melting (Tm): An endothermic peak where a crystalline material absorbs heat to transition from solid to liquid.
- Curing Reactions: An exothermic process where thermosetting polymers or adhesives crosslink, releasing heat as new bonds form.
- Decomposition: The thermal breakdown of a material, which can be endothermic or exothermic.
- Oxidation: An exothermic reaction with oxygen, measured as Oxidative Induction Time (OIT) or Onset Temperature (OOT) to assess stability.
- Polymorphic Transitions: Transitions between different crystalline forms (polymorphs), critical in pharmaceuticals.
- Vaporization and Sublimation: Endothermic processes where a material turns into a gas.
Extracting Quantitative Data from a DSC Curve
Beyond identifying events, DSC analysis provides precise quantitative data:
- Enthalpy (ΔH): Calculated by integrating the area under a peak (e.g., melting or crystallization), this value quantifies the energy of a transition.
- Degree of Crystallinity: For polymers, this is calculated by comparing the measured enthalpy of fusion to the value for a 100% crystalline sample of the same material.
- Specific Heat Capacity (Cp): Determined from the baseline of the thermogram. The step change at Tg allows for calculating the change in heat capacity (ΔCp).
- Purity Determination: For pure compounds, impurities cause a broadening and depression of the melting peak. The van’t Hoff equation can be applied to the peak shape to estimate purity.
- Transition Temperatures (Tg, Tc, Tm): Precisely determined from onsets and peak maxima, these are fundamental properties for material selection and processing.
The Power of DSC Analysis in Industry and Research
The insights from DSC analysis are critical for material characterization, quality control (QC), R&D, and process optimization across numerous industries. From aerospace to pharmaceuticals, DSC provides indispensable data for product performance and safety.
Key Applications of DSC Analysis
DSC analysis is remarkably versatile, with key applications in:
- Polymers and Plastics: DSC is essential for determining a polymer’s glass transition (Tg), melting point (Tm), and crystallinity, which dictate properties like flexibility, strength, and clarity. It’s also used to analyze thermal history, blend miscibility, and curing reactions in thermosets.
- Pharmaceuticals and Biologics: In this regulated sector, DSC is used to identify polymorphs of active ingredients, assess purity, and monitor stability. For biologics, it is invaluable for studying protein unfolding and thermal stability, which is critical for formulation and shelf-life prediction, as highlighted in reviews of DSC applications in biology and nanoscience.
- Food Science: DSC helps optimize food processing by studying starch gelatinization, the melting/crystallization of fats (e.g., in chocolate), and protein denaturation in dairy and meat. It can also assess the impact of processing on food components.
- Advanced Materials: In fields like nanoscience, DSC characterizes nanocomposites, phase transitions in metals and ceramics, and the thermal stability of novel compounds. It helps quantify amorphous content and study the behavior of nanostructured materials.
Understanding Material Stability, Purity, and Kinetics
DSC provides a powerful lens for understanding fundamental material behaviors:
- Material Stability: DSC assesses thermal stability by identifying decomposition temperatures. Specialized tests like Oxidative Induction Time (OIT) and Oxidative Onset Temperature (OOT) measure a material’s resistance to oxidation, which is crucial for predicting the shelf-life of polymers, foods, and pharmaceuticals.
- Purity Analysis: DSC offers a rapid method for estimating the purity of crystalline compounds. By analyzing the melting peak depression caused by impurities and applying the van’t Hoff equation, it provides a quantitative measure of purity.
- Reaction Kinetics: DSC is excellent for monitoring heat-related chemical reactions like curing or polymerization. By tracking heat flow over time, it can determine reaction rates, activation energies, and the degree of cure, enabling process optimization for products like adhesives and composites.
Achieving Precision: Best Practices and Advanced Techniques
Obtaining reliable and accurate data from DSC analysis requires careful experimental design and control. Meticulous attention to detail ensures the highest level of precision and interpretation.
Experimental Considerations for Reliable Data
To ensure the best possible DSC analysis results, several experimental factors must be carefully controlled:
- Sample Preparation: A small sample mass (1-20 mg) is ideal to avoid thermal gradients. Good thermal contact between the sample (solid, powder, film, or liquid) and the crucible is crucial.
- Crucible Selection: The choice of crucible is critical. Aluminum is common for general use, while platinum or gold is used for higher temperatures. Lids can be crimped for solids, hermetic (sealed) for volatile samples, or vented to allow gases to escape.
- Scan Rates: Heating/cooling rates (0.02 to 300 °C/min) affect the results. Faster rates increase sensitivity but can reduce resolution, while slower rates provide better resolution for thermodynamic calculations. A typical rate for safety screening is 2–3 °C/min.
- Purge Gas: An inert gas like nitrogen prevents unwanted oxidation and creates a stable atmosphere. For oxidative studies (OIT), a switch to oxygen or air is performed.
- Instrument Calibration: Rigorous calibration for temperature and heat flow using certified reference materials (e.g., Indium, Zinc) is essential. Advanced methods like Tzero calibration correct for instrument imbalances, significantly improving heat flow accuracy.
- Relevant Standards: Adherence to international standards like ASTM E967, ASTM E968, and ISO 11357 ensures data is comparable and reliable.
Advanced DSC Techniques
Beyond conventional DSC, advanced techniques offer deeper insights:
- Modulated DSC (MDSC): By superimposing a sinusoidal temperature oscillation on the linear heating rate, MDSC separates the heat flow signal into its reversing (thermodynamic, e.g., Tg) and non-reversing (kinetic, e.g., curing) components. This is excellent for resolving overlapping thermal events.
- Fast-Scan DSC (FSC): This technique uses ultra-high heating and cooling rates (e.g., >600 °C/min) to “trap” kinetic processes like crystallization or degradation. It is invaluable for studying unstable materials and improves sensitivity for weak transitions.
- Pressure DSC (PDSC): Performing DSC analysis under liftd pressure is useful for accelerating oxidative stability tests (OIT), suppressing the boiling of solvents, and studying pressure-dependent reactions.
- Photo-DSC (UV-DSC): This variant integrates a UV light source to study light-initiated reactions, such as the curing of dental resins or coatings, by monitoring the heat released during photopolymerization.
These advanced techniques, often coupled with traditional DSC, provide a comprehensive suite of tools for understanding the most complex material behaviors.
Frequently Asked Questions about DSC Analysis
What is the main output of a DSC analysis?
The main output of a DSC analysis is a thermogram, which is a graphical representation plotting the differential heat flow (typically in milliwatts, mW, or normalized per milligram, mW/mg) on the Y-axis against temperature (in degrees Celsius, °C, or Kelvin, K) or time (in minutes, min) on the X-axis. This curve reveals all the thermal transitions that occur within the material as it is heated, cooled, or held isothermally. It’s like a thermal “heartbeat” of your material, showing when it absorbs or releases energy.
How much sample is needed for a DSC test?
For most DSC analysis applications, a relatively small amount of sample is needed. Typically, we use between 1 and 20 milligrams. The exact mass can vary depending on the material’s nature, the expected magnitude of the thermal transitions (e.g., highly energetic transitions might require less sample), and the specific type of DSC instrument being used. For instance, some specialized micro-DSC units can work with even smaller, nanogram quantities. This small sample requirement is a significant advantage, especially when working with precious or limited materials.
Can DSC determine the composition of a material?
DSC analysis does not directly determine the elemental or chemical composition of a material in the way techniques like spectroscopy or chromatography do. However, it identifies and quantifies thermal transitions that are characteristic of a material’s physical and chemical structure. This data can be used to infer information about composition. For example:
- By measuring the enthalpy of fusion, we can calculate the percentage of crystallinity in a polymer, which is a compositional characteristic.
- Observing specific melting points can help identify known components in a mixture.
- Purity determination through freezing-point depression provides a quantitative measure of impurities, which is indirectly related to the material’s overall composition.
- The presence or absence of specific glass transitions can indicate the blending of different polymers or the presence of plasticizers.
So, while not a direct compositional analysis, DSC provides invaluable indirect evidence and quantitative data that contribute significantly to understanding a material’s makeup and behavior.
Conclusion
As we’ve explored, Differential Scanning Calorimetry (DSC) analysis is an indispensable and remarkably versatile thermal analysis technique. From the fundamental principles of heat flow measurement to the intricacies of advanced modulated and fast-scan methods, DSC provides a comprehensive thermal fingerprint of materials. It allows us to precisely identify and quantify critical thermal events like glass transitions, melting, crystallization, curing, and decomposition, offering insights into material stability, purity, and reaction kinetics.
The power of DSC analysis lies in its ability to translate these thermal behaviors into actionable data for diverse industries. Whether it’s optimizing polymer formulations, ensuring the stability and purity of pharmaceuticals, perfecting food textures, or advancing cutting-edge nanomaterials, DSC plays a pivotal role. It helps us predict material performance, troubleshoot issues, ensure quality, and drive innovation from research and development through manufacturing and quality control.
For expert DSC analysis and data interpretation, partner with a dedicated lab like Materials Metric. We provide high-precision analytical testing, materials characterization, and scientific consulting, delivering ISO 9001:2015-aligned data and expert interpretation to open up deeper insights into your materials and drive innovation across your projects. Contact us today to explore how our thermal analysis services can support your specific requirements.



